
by Lauren Richardson,
Sustainability Associate at Green Badger
GBCI Pro Reviewer
Health Product Declarations: the goals, benefits, and uses of HPDs and how they relate to LEED v4.
The HPD Open Standard is a voluntary, technical specification for reporting information on product contents and related health information and an evolving standard that will incorporate reporting for new health-related data as it becomes available. HPDs are technical documents. Their main purpose is to provide accurate and consistent information about the chemical contents and health-related information of building products.

The Objective of Health Product Declarations
Who created the HPD Open Standard?
A coalition of architects, designers, specifiers, owners, manufacturers, contractors, materials researchers, and non-governmental organizations (NGOs).
What are the goals of the HPD?
- to offer more transparency and disclosure about human and environmental health aspects of building products
- to create a standard for how to report information about product contents and their related health information
- to encourage voluntary product innovation and collaboration among product manufacturers.
What are the benefits of HPDs?
- information for better product selection and setting goals for materials selection
- awareness of human and environmental health (potential hazards)
- flexibility to disclose information to the level a manufacturer chooses (from minimum requirements to full disclosure/ transparency)
- a common language and database for improving and optimizing products
- a screen for substances identified as priority chemicals of concern
- selection of alternative products in order to avoid priority chemicals of concern
- comparison of specific products with generic products
What are the limitations of HPDs?
- It is not a certification or eco-label.
- It is not an assessment of product performance.
- It is not a life cycle assessment.
- It does not identify whether a product is “better” or “worse” than another.
- There are not enough products with HPDs to allow content and hazard information to be used consistently.
Who is responsible for creating HPDs?
The product manufacturer or a third- party preparer, on behalf of the manufacturer, creates the HPD.
Who manages HPD Development?
- The HPD Collaborative (HPDC), is a nonprofit member organization representing a cross-section of the building industry participants manages HPD development.
What makes an HPD a “good HPD?”
- The HPD Open Standard gives manufacturers a standardized way of reporting at higher levels of disclosure – and more stringent thresholds – than may be required for a given certification.
- HPDs can also provide inventory and hazard information to feed into Declare, Cradle to Cradle and Mindful Materials
- HPDs can also be used as documentation for credits in the LEED rating system and the WELL Building Standard
Why HPDs are important for human health, and how they can contribute to healthier buildings:
Project teams without special knowledge in material health can use HPDs to:
- Achieve requirements of certification systems, such as LEED v4 and the WELL Building Standard.
- Inform product selection in conjunction with other resources, such as priority chemicals lists, compare the health-related attributes of products.
- Collaborate with manufacturers on product innovation
- Send an important signal to manufacturers. High profile design teams write letters to manufacturers letting them know they will not specify products without HPDs.
Creating HPDs is time consuming and expensive, so why do manufacturers continue to make HPDs?
- Manufacturers are rewarded for products that offer disclosure by increased specification of those products
- Manufacturers are further reward when they make improvements in the health attributes of their products
- This feedback encourages manufacturers to do the same for more of their products.
- Asking for HPDs that do not exist also sends messages to manufacturers that design teams place a priority on products with HPDs.
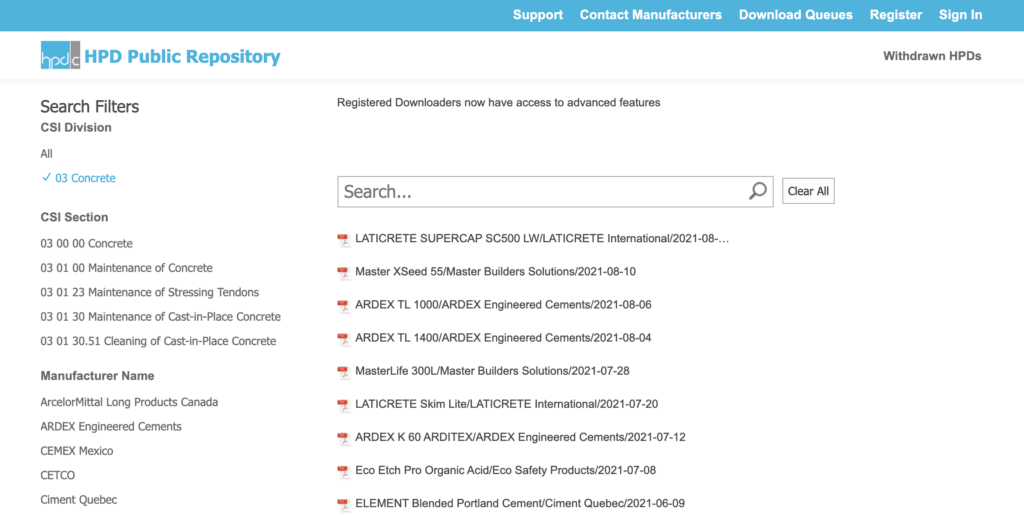
Finding Health Product Declarations
- is an online resource that is free for users
- is where manufacturers to publish their HPDs
- once published there, HPDs may also appear on manufacturer websites and other product libraries and databases.
- can be searched by CSI division/section, manufacturer name, product name, and LEED v4 Pre-Check status (both Options 1 and 2).
NOTE: HPDs included in the HPDC Repository are not reviewed or verified by HPDC. However, an HPD not appearing on the HPD Public Repository is not officially published, and not valid for LEED credit.
Part 2: How to Read an HPD with Health Product Declaration Example
In addition, this section will help you understand the HPD format and understand what constitutes a “compliant” HPD. Interpret the information presented in an HPD and appreciate how HPD can be used by design professionals.
There are six sections of a health product declaration:
- Section 1: Summary – highlights the contents of the product and their hazards, as well as key reporting elements such as thresholds
- Section 2: Content in Descending Order of Quantity – This section is the heart of the HPD. It lists contents in the product consistent with established thresholds (amounts), their associated hazards, and other relevant information
- Section 3: Certifications and Compliance – for example, certifications for VOC content and emissions
- Section 4: Accessories – for example, what’s needed to install or maintain the product
- Section 5: General Notes – for example, explanations from the manufacturer
- Section 6: References – for example, links to hazard lists used in the HPD
Health Product Declaration Example Breakdown
Section 1: Summary
Materials and Substances
- Material — a uniform solid, liquid, or gas. Materials are composed of one or more “substances.”
- Substance — molecules, atoms, etc. and their properties, such as density, melting point, etc.
- Content — refers to both materials and substances more generally
Products can be made up of just a few materials or be very complex, involving scads of materials and lengthy supply chains.

Health Product Declaration Thresholds
Threshold – the amount of a substance that must be present for that substance to be reported,
Thresholds can be reported as:
- Parts per million (ppm)
- a threshold of 100 ppm (0.01%) is more stringent than a threshold of 1,000 ppm (0.1%); it is likely to lead to disclosure of more substances.
Thresholds can apply to the whole product or to each material in the product (for example, 1,000 ppm for some materials and 100 ppm for others.)

Health Product Declarations: Characterized, Screened, Identified
- “Characterized” — whether the percent weight (quantity), role or function are provided for all substances above the threshold indicated.
- “Screened” — whether all substances above the threshold were screened for hazards using lists in the HPD Open Standard — and results — are reported.
- “Identified” — whether all substances above the threshold are reported by name
This reporting flexibility gives manufacturers the ability to withhold information about substances as a way to protect proprietary information while still disclosing potential hazards.
This type of reporting is compliant with the LEED v4 Material Ingredients credit.
“Yes Ex/SC” means that the requirements to Characterize, Screen, and/or Identify have been met using HPDC-approved Special Conditions guidance for specific classes of materials. This indicates compliance with LEED v4.
- Contents’ highest concern GreenScreen Benchmark or List Translator Score. This section reports on the most hazardous GreenScreen List Translator or Benchmark Score found in any of the substances in the product.
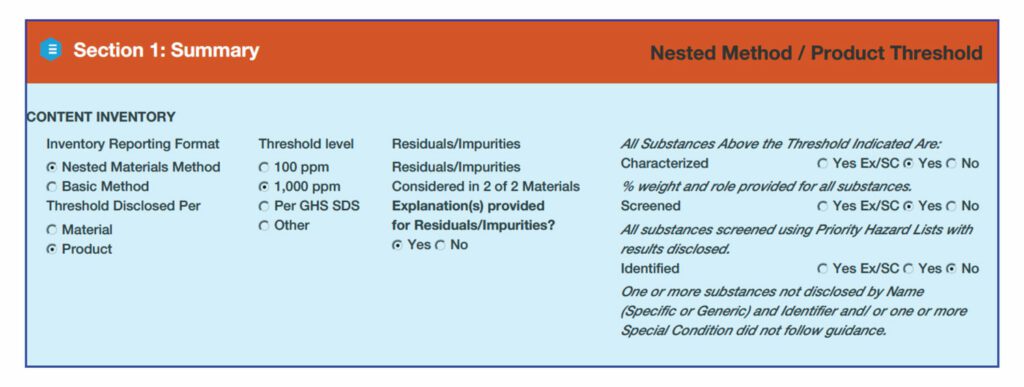
HPD Content Inventory: Inventory Reporting Format
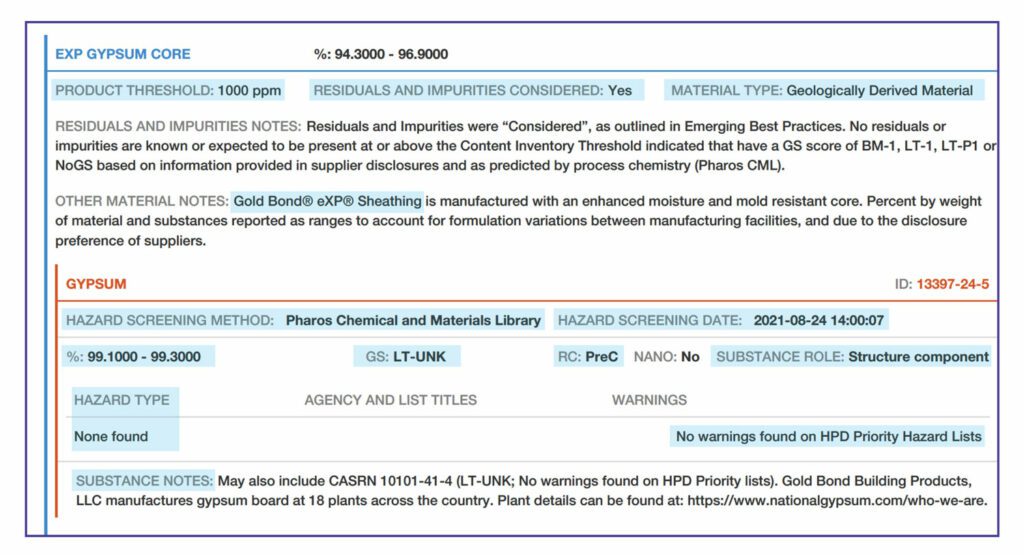
There are three methods for creating a content inventory, based on how contents are categorized and how reporting thresholds are applied.
Nested Materials Inventory Method: contents are reported first at the materials level. Substances are then itemized within each material
“Basic” Inventory method. Materials are not identified. This means that there will be a single list of all substances within the product.
- Residuals and Impurities: trace amounts of substances remaining after the manufacturing process that were not intentionally added for a functional purpose in the final product, defined as follows:
- Residual: a substance left over from a prior process, such as a residual monomer in a polymer from incomplete polymerization, that is not intended to remain in the final product
- Impurity: a substance that was present in the raw material, such as asbestos that often is associated with vermiculite.
- “Considered” means that they have made an effort to identify the residuals and impurities
- “Not Considered” means no effort has been made to identify residuals and impurities; the manufacturer must explain why
- Characterized, Screened, Identified. This section summarizes the completeness and type of disclosure in the HPD.
- GreenScreen for Safer Chemicals scores. GreenScreen® for Safer Chemicals is a program of the nonprofit organization Clean Production Action. It facilitates comparative chemical hazard assessment at the substance level. There are two types of GreenScreen results that can be reported in an HPD:
- List Translator scores that are produced through an automated tool that screens against specified hazard lists. No assessment is performed. For more information on the GreenScreen List Translator™: https://www. greenscreenchemicals.org/method/greenscreen-list-translator
Benchmark scores represent a more comprehensive assessment than List Translator scores, developing hazard levels for a full set of environmental and human health endpoints.

The HPD summary includes:
- Number of GreenScreen BM-4/BM-3 Contents. This section reports on the total number of substances with lower hazards – Benchmark-3 (use, but there’s still opportunity for improvement) or Benchmark-4 (preferred, this is a safer chemical).
- Contents’ highest concern GreenScreen Benchmark or List Translator Score. This section reports on the most hazardous GreenScreen List Translator or Benchmark score found in any of the substances in the product.
Nanomaterials. – substances that have been engineered to be very small (down to 10,000 times smaller than the diameter of a human hair). The impact of types of nanomaterials on the environment and human health are still unclear.
Inventory and Screening Notes – explanation from the manufacturer– for example, why residuals and impurities were not considered.
Section 2: Content in Descending Order of Quantity

- Nested Materials Inventory: each material is listed with its substances, associated hazards, and GreenScreen scores – all in abbreviated form.
- Basic Inventory: substances are listed for the product as a whole.
Volatile Organic Compound (VOC) Content. VOC content information must be provided for all liquid/wet-applied products.
- “Exempt VOCs” – these VOCs do not contribute to smog formation and hence are exempted from outdoor air regulatory reporting. However, some of the exempt VOCs still present health hazards.
- “Material (g/L)” – the amount of non-exempt VOC content compared to the volume of the entire product in grams per liter.
- “Regulatory (g/L)” – the amount of non-exempt VOC content compared only to the volume of the non- exempt content in the product in grams per liter. (This measure is used by regulatory agencies.)
- “Ultra-low VOC tints” – for tintable products, are ultra-low VOC tints available? “yes” and “no,” and “NA” means that the product is not tintable.
Certifications and Compliance. This section lists certifications that are detailed in Section 3 of the Format. When applicable, VOC emissions and VOC content certifications are included here. Other certifications related to content (materials and substances) and health (hazards, etc.) may be included.
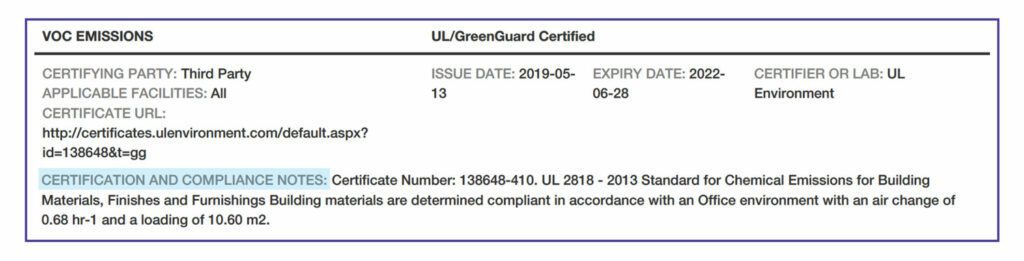
Third Party Verification. Third party verification involves audits to check aspects of the HPD.
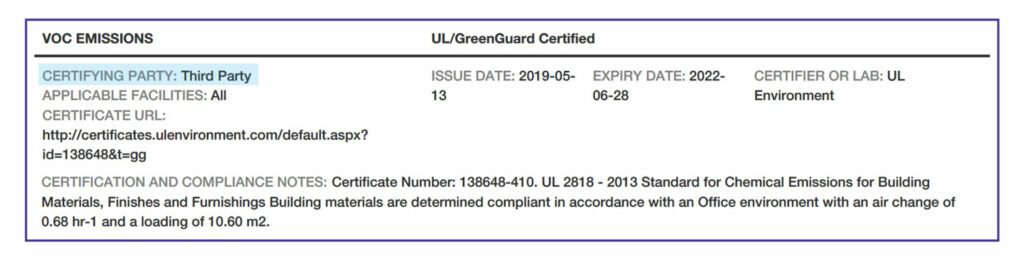
Preparer and Verification. Third parties can provide expertise in gathering and analyzing data as well as protect confidential information.
Screening, Published, and Expiration Dates. HPDs expire three years after the screening date. HPDs uploaded to the HPDC Public Repository are considered “published.”
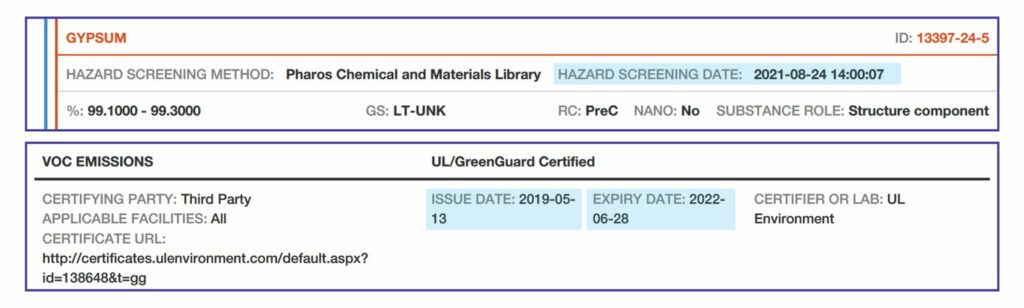
Substance Name. The manufacturer can provide a specific name, generic name or chemical class. “Undisclosed” means that the manufacturer chooses not to disclose the specific name of the substance, and the reason must be explained in the Notes
“Undisclosed” substances cannot be considered “identified”.
“Unknown” means that the manufacturer does not know the substance name, due to lack of disclosure by the supplier.
Identifier. The primary identifier is the Chemical Abstract Service Registration Number (CAS RN). Other identifiers can be used when there is no CAS RN. The CAS RN cross-references with the hazard lists referenced in the HPD Open Standard and provides precise identification.
Percent (%).
- Nested Materials Inventory method. The percent by weight of the substance in the material.
- Basic Inventory method. The percent by weight of the substance in the product.
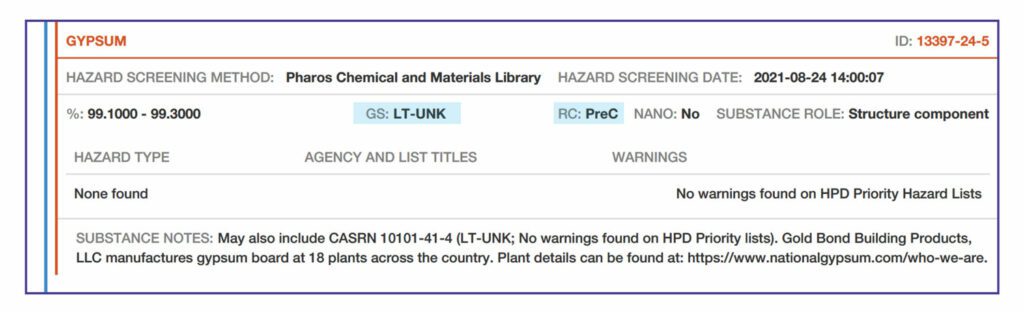
GreenScreen (GS). See Section 1 – “Number of lower hazard substances in Product” and “Most hazardous substance score found in Product” for explanation of GreenScreen® scores.
Recycled Content (RC). Indicates whether the substance has recycled content.
- “PostC” indicates post-consumer recycled content.
- “PreC” indicates pre-consumer (post-industrial) recycled content.
- “Both” indicates both are included.
- “None” indicates no recycled content.
- “UNK” indicates that use of recycled content is unknown.
Substance Role – the substance’s purpose in the material, such as binder, antimicrobial, flame retardant, preservative, etc.
Hazard Screening – provides data about whether a particular chemical (a “substance”) has been associated with one or more health hazards
Determining exposure and risk requires additional product assessment.
The GreenScreen approach makes it possible to compare chemical hazards. There are two types of GreenScreen results that can be reported in an HPD:
- List Translator(LT) scores: produced through an automated tool that screens against hazard lists specified in the HPD Open Standard. No assessment is performed. For more information: https://www.greenscreenchemicals.org/method/greenscreen-list-translator
- Benchmark (BM) scores: produced through a Certified Full GreenScreen Method assessment that includes a toxicological review of all available information from scientific studies, hazard lists and modelling or analogs. For more information: https://www.greenscreenchemicals.org/method/full- greenscreen-method
Benchmark scores are a more comprehensive assessment than List Translator scores, as they develop hazard levels for a full set of environmental and human health endpoints.
In the HPD, each substance is screened against Priority Hazard Lists to see if the substance is listed.
If it is listed, the hazard type (cancer, respiratory, physical, etc.) , is recorded along with the Agency List Abbreviation (CA EPA, IARC, etc.) and Hazard Warning (carcinogenic to humans, asthmagen, explosive, etc.)
Lists and agencies can be found in the HPD Open Standard, Appendix D.
Lists were selected for inclusion primarily based on criteria developed for GreenScreen. All lists in the GreenScreen List Translator are included if they contain high or very high hazard scores
Additional lists have been included to address ozone depletion, global warming, brominated and chlorinated flame retardants, and other chemicals.
HPDC’s online HPD Builder cross-references these lists based on name and/or CAS RN.
Substance Notes. This section includes information to further explain the entries above.
Section 3: Certifications and Compliance
This section lists independent certifications that relate to contents and health-related characteristics.
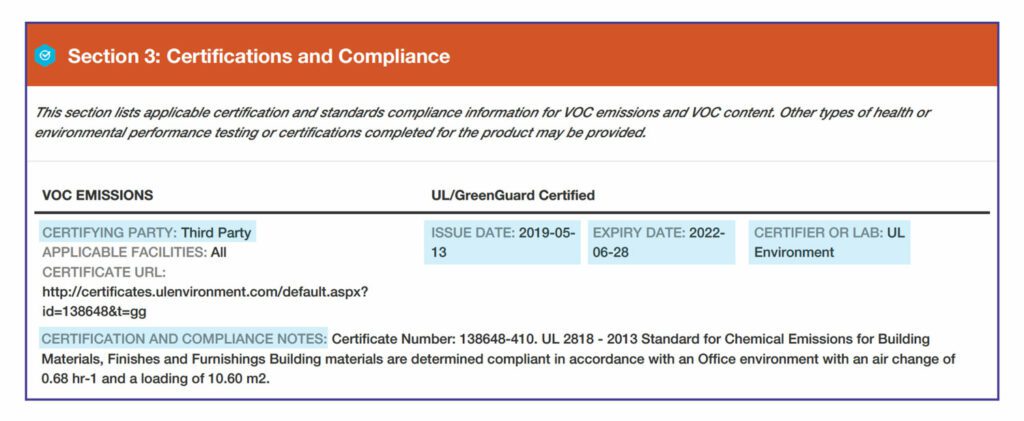
VOC Emissions. There are two methods for this testing: California Department of Public Health Standard Method and the international AgBB standard.
Programs that certify VOC emissions with the California standard include Indoor Advantage Gold™, FloorScore®, and GREENGUARD Gold. Some products have no accepted emissions testing , so “N/A” is indicated. If the manufacturer has chosen not to test or certify the product, “not tested” is indicated. Or, a product might be indicated as “inherently non-emitting,” such as tile.
VOC Compliance. All products that are wet-applied on site, such as paints, adhesives, or other coatings, must have an entry noting the certification or applicable standard with which the product complies.
Applicable Facilities. The specific manufacturing facilities that are covered by the certifications and standards noted above.
Section 4: Accessories

This section lists additional products that must be used for installation or for warranty compliance (like adhesives, fasteners, field coatings). Products that are recommended but not required for installation, maintenance, cleaning, or operations can be included. These products can contain hazards. If there is an HPD for the accessory, a URL link is provided.
Section 5: General Notes
Several sections in the Standard require notes, which can be specific to a material or substance, or general.

Manufacturer Information. This section provides contact and other basic information on the manufacturer.
Key to Acronyms, Abbreviations, and Terms. This section contains a key to acronyms, abbreviations, and terms used in the document
How products with an HPD can contribute to points in LEED v4 and v4.1:
For more information about one way to earn the Material Ingredient reporting credit (which is Option 1 in Material Ingredients) in LEED versions 4 and 4.1 by using products with Health Product Declarations (HPDs) in this post. Additional sources you might investigate for health-related information on building products are buildinggreen.com, healthybuilding.net, and mindfulmaterials.com.






